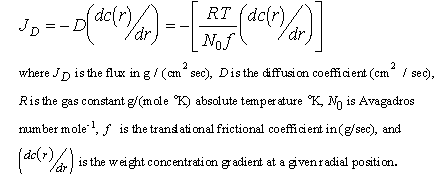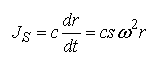Overview of Information Needed
Overview of Information needed:
Regardless of whether velocity or equilibrium experiments are conducted, the values obtained by direct analysis of the data must be adjusted for the particular conditions of the experiment. To demonstrate how the parameters of interest are determined from experimental data, the "working" equations are presented. For diffusion, Fick's first law for a single ideal component may be written using the Einstein-Sutherland approximation: (Ref. 18)
Equation 1
A measurement of D is sufficient to calculate f, and methods for doing this from sedimentation data are described elsewhere. (Ref. 9 10 11)
The flux due to sedimentation in a two component system is the product of the mass concentration of solute and its velocity:
Equation 2
where c is the mass concentration of solute particles, dr/dt is their velocity (cm/sec), s is the sedimentation coefficient (sec), ω^2 is the square of the rotor's angular velocity and r is the distance from the center of rotation. The substitution of for dr/dt is appropriate since s is defined as the velocity of a particle divided by the acceleration (ω^2)*r . (note: There are limitations to this substitution for solutions that exhibit highly non-Newtonian viscosity.) As will be shown in the next section, it is the measurable parameter s that is of immediate interest. (Ref. 20 21 22 23 24)